Research & development
With the help of its technology platform, Xintela is developing new treatments in the fields of stem cell therapy and targeted cancer therapy, with focus on indications with a high unmet medical need.
Marker technology
Integrins are a family of cell surface proteins that regulate important cellular functions. Some integrins have long been used as target molecules in the development of therapies for inflammatory diseases and cancer, among others. Xintela’s research and development uses integrin α10β1, which was discovered by Evy Lundgren-Åkerlund’s research group at Lund University. Lundgren-Åkerlund and colleagues have shown that integrin α10β1 is found on cartilage cells and on mesenchymal stem cells (MSCs) that can develop into different cell types, including cartilage cells. This discovery forms the basis for Xintela’s process for the selection of MSCs using integrin a10b1 and the development of the stem cell products XSTEM® and EQSTEM®.
Stem cell therapies
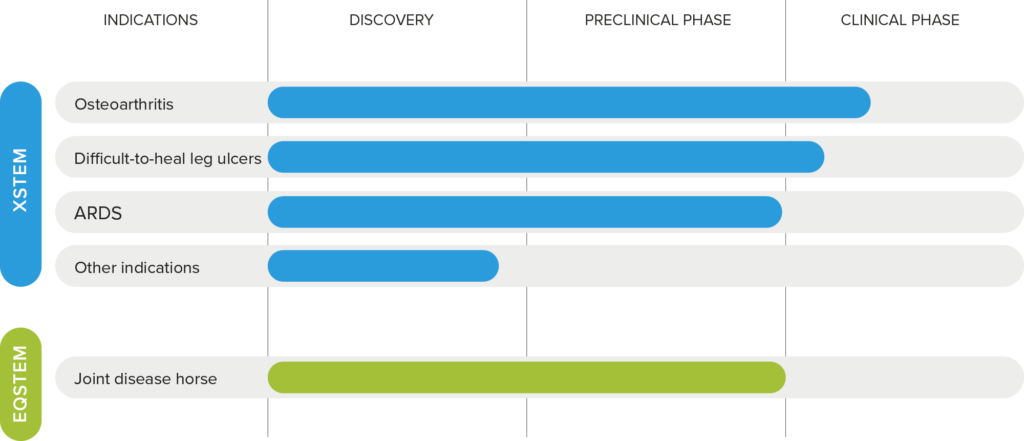
In stem cell therapy, the stem cell marker integrin α10β1 is used to select and quality assure stem cells in the patented stem cell products XSTEM®, for the treatment of humans, and EQSTEM®, for the treatment of horses. Xintela has successfully conducted clinical studies (phase I/IIa) with XSTEM® for the treatment of knee osteoarthritis and difficult-to-heal venous leg ulcers and preclinical studies for the treatment of ARDS (Acute Respiratory Distress Syndrome) as well as preclinical studies with EQSTEM® in horses with osteoarthritis.
XSTEM®
The stem cell product XSTEM®
XSTEM® consists of allogeneic (donated) integrin α10β1-selected and quality-assured mesenchymal stem cells (MSCs) from adipose tissue from healthy adult donors. XSTEM® is patented both as a product and for therapeutic use in all indications. This gives Xintela a unique position to develop safe and effective stem cell-based treatments for a large number of diseases that currently lack effective treatment.
Own GMP-facility
Xintela’s stem cell products are produced in the company’s own GMP-approved facility, which significantly reduces both production costs and the capacity and scheduling risks associated with contracting out.
XSTEM® clinical study for the treatment of knee osteoarthritis
Xintela has reported excellent 24-month results from our clinical phase I/IIa study in knee osteoarthritis where three different dose levels of XSTEM® (4, 8 and 16 million stem cells) were evaluated in a total of 24 patients (8 patients/dose level). The results show that XSTEM® is safe and that the treatment provides a significant and clinically relevant reduction in knee pain and improved joint function as well as improved cartilage and bone structure, confirming a long-lasting treatment effect and disease-modifying potential.
Link to press release about the study »
Read more about osteoarthritis » below
XSTEM® clinical study for the treatment of difficult-to-heal leg ulcers
In Xintela’s clinical study (phase I/IIa) in patients with difficult-to-heal venous leg ulcers, six patients received treatment with XSTEM® or placebo applied to the wound and followed weekly for ten weeks and at four months after dosing. The primary goal of the study was to demonstrate that the treatment is safe and tolerable. The final report from the study will be published in May 2026.
Read more about difficult-to-heal leg ulcers » below
XSTEM® for Acute Respiratory Distress Syndrome (ARDS)
Xintela has successfully conducted preclinical studies for the treatment of ARDS with XSTEM® in collaboration with Skåne University Hospital. The results showed that XSTEM® reduced blood coagulation and damage to lung tissue. These results may eventually have application in, for example, sepsis where approximately 30% of patients develop ARDS with high mortality.
EQSTEM®
EQSTEM® for Equine Joint Disease
Based on the company’s unique stem cell selection technology, Xintela has developed the stem cell product EQSTEM® for horses. EQSTEM® has shown safety and efficacy in two osteoarthritis studies in horses. Xintela has entered into a collaboration and license agreement with EQGen Biomedical Inc. for the continued development and commercialization of EQSTEM® and stem cells for other animals.
Link to publication (Delco) »
Link to publication Andersen) »
Targeted cancer therapy
Integrin α10β1 – Target molecule on aggressive cancer cells
Xintela’s research team discovered that integrin α10β1 is expressed by aggressive and difficult-to-treat cancers such as triple-negative breast cancer, glioblastoma and lung cancer. This led to Xintela’s investment in the development of therapeutic antibodies and ADCs (antibody drug conjugates) targeting integrin α10β1. Our oncology business is run by our wholly-owned subsidiary Targinta.
Indications
Osteoarthritis
Osteoarthritis is a joint disease characterized by degradation of the articular cartilage and impaired function of the cartilage cells. It is the most common chronic joint disease, especially in the knees, hips and hands, as well as the most common cause of disability in the elderly. The main symptoms are severe pain, inflammation, stiffness in the joint and reduced mobility. The disease affects about 25 percent of all individuals over the age of 60 and is increasing in extent due to an increasing elderly population. Drugs offered today are primarily pain-relieving and anti-inflammatory, which treat the symptoms but not the actual cause of the disease.
Difficult-to-heal leg ulcers
Difficult-to-heal leg ulcers in the elderly, including venous leg ulcers, are a major medical problem that results in pain and reduced quality of life for the patient, as well as large costs for healthcare systems. The incidence increases with age and is estimated to be about 4 percent among people over 65 years of age. Today’s treatments for difficult-to-heal leg ulcers include compression techniques and various surgical techniques, but there is a lack of effective drugs.
Acute Respiratory Distress Syndrome (ARDS)
ARDS is a form of acute severe lung failure that can occur as a result of, for example, pneumonia, trauma and blood poisoning. The condition means that the lung function collapses and mortality is high. There is currently no effective treatment for ARDS.
Publications
Camper, L., Holmvall, K., Wängnerud, C., Aszódi, A., and Lundgren-Åkerlund, E. “Distribution of the collagen-binding integrin α10β1 during mouse development.” Cell Tissue Res. 306, 107-116 (2001).
Bengtsson, T., Camper, L., Schneller, M., and Lundgren-Åkerlund, E. “Characterization of the mouse integrin subunit α10 gene and comparison with its human homologue. Genomic structure, chromosomal localization and identification of splice variants.” Matrix Biology 20, 565–76 (2001).
Camper, L., Hellman, U., and Lundgren-Åkerlund, E. Isolation, “Cloning, and Sequence Analysis of the Integrin Subunit α10, a β1-associated Collagen Binding Integrin Expressed on Chondrocytes.” Journal of Biological Chemistry 273, 20383–20389 (1998).
Bengtsson, T., Aszódi. A., Nicolae, C., Hunziker, E.B., Lundgren-Åkerlund, E. and Fässler, R. “Loss of α10β1 integrin expression leads to moderate dysfunction of growth plate chondrocytes. “ Journal of Cell Science 118, 929–36 (2005).
Varas, L., Bryngelson Ohlsson, L., Honeth, G., Olsson, A., Bengtsson, T., Wiberg, C., Bockermann, R., Järnum, S., Richter, J., Pennigton, D., Johnstone, B. Lundgren-Åkerlund, E and Kjellman, C. “α10 Integrin expression is up-regulated on fibroblast growth factor-2-treated mesenchymal stem cells with improved chondrogenic differentiation potential. ” Stem Cells and Development 16, 965–978 (2007).
Lundgren-Åkerlund E., Aszòdi A. “Integrin α10β1: a collagen receptor critical in skeletal development” Adv
Exp Med Biol. 819:61-71. (2014)
Zeltz C, Lu N, Gullberg D. “Integrin α11β1: a major collagen receptor on fibroblastic cells” Adv Exp Med Biol. 819:73-83. (2014)
Munksgaard Thorén M., Chmielarska Masoumi K., Krona C., Huang X., Kundu S., Schmidt L., Forsberg-Nilsson K., Floyd Keep M., Englund E., Nelander S., Holmqvist B. and Lundgren-Åkerlund E. “Integrinα10,a Novel Therapeutic Target in Glioblastoma, Regulates Cell Migration,Proliferation, and Survival.” Cancers (Basel). 11, 587 (2019).
Uvebrant K., Reimer Rasmusson L., Talts JF., Alberton P., Aszodi A., and Lundgren-Åkerlund E. “Integrinα10β1-selected Equine MSCs have Improved Chondrogenic Differentiation, Immunomodulatory and Cartilage Adhesion Capacity.” Ann Stem Cell Res. 2,001–009 (2019).
Delco ML., Goodale M., Talts JF.,Pownder SL., Koff MF., Miller AD., Nixon B., Bonassar LJ., Lundgren-Åkerlund E., and Fortier LA. “Integrin α10β1-SelectedMesenchymal Stem Cells Mitigate the Progression of Osteoarthritis in an EquineTalar Impact Model.” Am J Sports Med. 48, 612-623 (2020).
Andersen, C., Uvebrant, K., Mori, Y. Aarsvold S., Jacobsen S., Berg LC., Lundgren Åkerlund, E., and Lindegaard C. ”Human integrin α10β1-selected mesenchymal stem cells home to cartilage defects in the rabbit knee and assume a chondrocyte-like phenotype.” Stem Cell Res Ther. 13, 206 (2022).
Clarke EJ., Johnson E., Caamano Gutierrez E., Andersen C, Berg LC., Jenkins RE., Lindegaard C., Uvebrant K., Lundgren-Åkerlund E., Turlo A., James V., Jacobsen S., and Peffers MJ. “Temporal extracellular vesicle protein changes following intraarticular treatment with integrin α10β1-selected mesenchymal stem cells in equine osteoarthritis.” Front Vet Sci. 9:1057667 (2022).
Edström D., Niroomand A., Stenlo M., Uvebrant K., Bölükbas DA., Hirdman G., Broberg E., Lim HC., Hyllén S., Lundgren-Åkerlund E., Pierre L., Olm F., Lindstedt S. “Integrin α10β1-selected mesenchymal stem cells reduced hypercoagulopathy in a porcine model of acute respiratory distress syndrome.” Respir Res. 24(1):145 (2023).
Andersen C., Jacobsen S., Uvebrant K, Griffin, IV, JF., Vonk LA., Walters M., Berg LC., Lundgren-Åkerlund E., and Lindegaard C. “Integrinα10β1-Selected Mesenchymal Stem Cells ReducePain and Cartilage Degradation and Increase Immunomodulation in an EquineOsteoarthritis Model.” Cartilage. doi: 10.1177/19476035231209402 (2023).
Andersen C., Walters M., Bundgaard L., Berg LC., Vonk LA., Lundgren-Åkerlund E., Lyngfeldt Henriksen B., Lindegaard C., Skovgaard K., and Jacobsen S. “Intraarticular treatment with integrin α10β1-selected mesenchymal stem cells affects microRNA expression in experimental post-traumatic osteoarthritis in horses.” Front Vet Sci. 11:1374681 (2024).